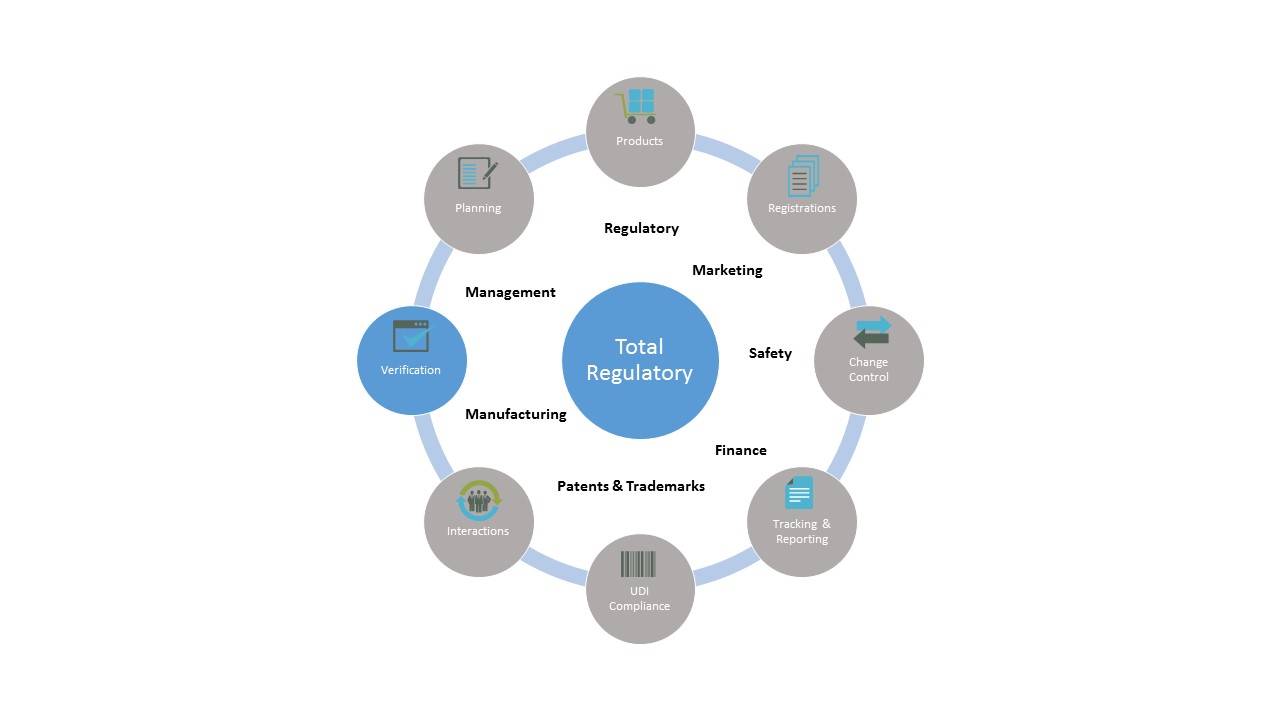
Product Registration Tracking with 123Compliance software enables medical device companies to significantly reduce efforts, costs and risks associated with the registration process by:
- Improving the management of global registrations
- Tracking submission activities
- Reporting capabilities to identify trends
Learn how product registration tracking with 123Compliance can help your organization quickly start or expand your automated regulatory affairs programs.